Introduction
In our modern world of 2017, nearly everyone uses cell phones. We use them to communicate, we use them to play games, we use them to get the latest news, we depend on them for the most basic of our day-to-day activities. But have you ever wondered how these mini-machines just keep going for years and years with the battery life close to brand new? Have you ever wondered why unlike other electronics, these tiny machines don’t burn down your house when you leave them plugged into a socket overnight? Well, you’re about to find out. This article dives deep into the world of lithium-ion batteries and digs on charging processes employed by phone companies to ensure optimum life span of smartphone batteries. This project explores the control system employed by most modern day cell phones, which has been vital to improving battery life, preventing overcharging (and its consequences), and saving our batteries from sudden death.
What exactly is controlled and why?
As complex as it may seem, a lithium-ion battery simply converts chemical energy to electrical energy by the movement of lithium ions from a negative electrode to a positive electrode. The number of ions available at the negative electrode is an indication of the energy available in the battery for use, and it is the very variable sought to be controlled (increased) by chargers. It is quantified as the “state of charge” by phone manufacturers and displayed in percentage on the phone screen.
Theoretically, and in reality, the amount of ions at the negative electrode could go to absolute limits of 0% and 100%, however such repeated cycles damage the battery. By letting the amount of ions in the electrodes go to absolute limits repeatedly, the structure of the electrodes degrades with time, and this reduces the amount of ions each electrodes can accept, and therefore reduces the battery capacity. To prevent the above, most phone systems display non-absolute limits within 10% of the absolute limits for the state of charge. In other words, a phone battery displaying 0% still has some ions at the negative electrode and could go less than 0% if it weren’t designed otherwise, and a phone battery displaying 100% can still accept ions at the positive electrode and go above 100% if the voltage limit was higher.
Controlling the state of charge while charging a battery is critical to its operation because, it ensures that the battery stays within usable percentage and that long-term battery capacity is conserved. Also by controlling the state of charge, overcharging and total battery depletion are prevented. Overcharging occurs when the battery is fed more voltage than it can handle in its charging cycle, and as a result, heat is produced. While other types of batteries, like lead acid batteries, can handle this slightly better, in lithium ion batteries it causes undesirable side reactions, such as plating out of the lithium metal. When this metal plates out, there are less of the titular “lithium ions” left to hold the charge, and as such, the battery loses capacity over time.
How does this system manipulate things to achieve control?
The controlled variable, the state of charge of a lithium-ion battery, is dependent on the number of ions at the negative electrode, and the number of ions at the negative electrode depends mainly the rate of discharge/charge of the battery (manipulated variable). As long as a phone is on, there will always be some discharge of the battery, and this rate of discharge would depend on the level of phone usage. Now, phone producers could have unsuccessfully tried manipulating the rate of discharge by advising users to use their phones intermittently, but instead phone manufacturers wisely chose to manipulate the rate of charge using wonderful innovations, called phone chargers.
As “complex” as phone chargers look, those portable things simply convert high voltage AC (alternating current) to low voltage DC (direct current), which is then suitable for and supplied to the battery. This step-down conversion is necessary because otherwise: BOOM!

Charging a phone is the exact opposite of discharging a battery’s energy. By supplying current (the manipulated variable), a charger transfers lithium ions from the positive electrode to the negative electrode, thus restoring energy. Now, if charging simply occurred in a non-controlled manner by providing a constant current flow to a phone, we’d all have two choices: be negligent and accidentally leave the phone charging for long periods (this would only overcharge the battery and eventually blow up our lovely homes), or we could be very watchful and constantly watch until when our phone gets to a 100% state of charge (who has time for this?). Lucky enough for us, cell phones are designed to be charged smarter today; the process of charging smartphones occurs in three stages. The first stage occurs rapidly; a steady current flow is taken in to ensure the charge rate is far higher than the discharge rate. As the state of charge in the battery increases to about 80% the smart phone switches to the second stage in which the voltage is relatively constant, but resistance is applied to current flow to decrease it until a 100% state of charge of the battery is achieved. Once a 100% state of charge is achieved, the smartphone switches to the third “trickle charging” phase, in which the rate of current flow into the battery is just enough to balance the battery’s discharge rate. This way, the battery is maintained at 100% state of charge. The above control system regulates the state of charge of the battery by manipulating the flow of current into the phone battery and this is why we can leave our phones charging all night and they will stay at 100% when you wake up, not 90%, not 105%; the control system minimizes overcharging and saves battery life.
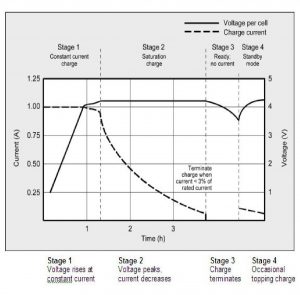
<http://batteryuniversity.com/learn/article/charging_lithium_ion_batteries>
What could possibly disturb the way my phone charges?
Disturbances to our control scheme come in several forms but the most common ones are an increased rate of battery energy depletion (when someone is charging their phone while using it) and changes to the amount of current coming into the charger from the socket. A simple charger can’t be expected to compensate for something like a blackout, but is able to compensate for increased usage rate, and small surges/depressions in the charge coming from the socket. For example, the charger is able to compensate for the sudden depression that comes from turning on another energy intensive appliance plugged into the same grid, like an air conditioner. It is also able to compensate for small surges, in the reverse situation of when the air conditioner is turned off, and there is suddenly a surplus of current coming through the socket. The charger, of course, shouldn’t be expected to compensate for something like a block-wide blackout, but small localized changes should be reasonable to manage. On the whole, though, changes to the current coming from the wall shouldn’t be commonplace, but the charger should be able to compensate for some small perturbations, as controlling the current going into the phone is the entire purpose of the charger’s control scheme.
In order to control the state of charge of the phone, the best way to do this is by implementing a straightforward feedback loop. In common vernacular, a feedback loop is a reactive model which involves measuring the controlled variable, and changing the manipulated variable upstream. A basic example would be of that of someone taking a shower: when you feel cold water, you increase the temperature, and when it is too hot, you decrease the temperature. Either way, you are making a change based on what the final outcome of the process is, which is what we are doing here.
This feedback loop measures the state of charge of the battery to determine the rate at which current should be taken by the battery. This will keep the phone charged but will not allow it to overcharge. Having a different rate of charge come in from the socket would simply be compensated for in what rate of charge the charger delivers to the battery. Since the charger already changes the current going into the batter, compensating for small perturbations in the original current should not be difficult. The diagram below represents a sample feedback loop implemented during the charging process of a cellphone.
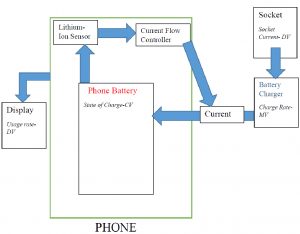
This above described process of cell phone charging is extremely important. It prevents overcharging without much human effort and conserves battery life. So hey! Next time you see a mobile engineer at Samsung, thank him for saving you time, saving you cost, and most importantly saving you from burns.
References:
http://www.batteryuniversity.com/
http://batteryuniversity.com/learn/article/charging_lithium_ion_batteries
http://www.apple.com/batteries/why-lithium-ion/
http://sciencing.com/smartphone-charger-stop-after-its-full-17317.html
Lithium-ion batteries, and general battery technology itself is often taken for granted by society today. We continue to neglect the awesome innovation that has occurred over years and years of study, which has allowed batteries to safely and efficiently power devices we use every day. I am thankful that you all have written about this subject matter because it not only has always excited me, but it is also important to learn how the devices you use daily operate. Obviously, we can quickly say controlling the rate of phone charging by manipulating the current supplied to a charger makes complete sense because it is a process that has been used for over a decade. Even though it was apparent, I was glad that the article further substantiated why this control scheme is needed since a phone would ultimately explode if it kept charging past 100%. However, it would have been nice if an explanation was given regarding why the phone would go “boom” if it overcharged.
At first when reading the first few descriptions about how the control scheme actually works I was a bit confused as to the actual process that goes on to allow a change in charge to manipulate the rate of charging. However, after seeing the diagram provided the process made complete sense. While I understand that when the cell phone charge percent is checked, a current flow controller decides whether to decrease the amount of current supplied to the phone or not, how the trickle charging works is less clear. I believe the article could have elaborated onto how the controller determines the exact discharge rate of the phone so it can match this discharge rate with the current it supplies to the phone. I was also hoping for some elaboration as to alternative control schemes that could be used instead of those that are currently implemented.
Overall, I enjoyed reading the article, mostly due to the fact that I am already interested in this specific subject material. I thought that the specific control process was explained accurately, and the use of a diagram made it even clearer. Since the specific control scheme is also already implemented in all phone chargers it is obvious that I can see it being used in practical applications. However, I feel the article should have elaborated on possible alternatives to the current charging control scheme, or maybe talked about how control schemes on wireless charging pads work (as nothing is plugged into a charging port for a sensor to make a decision). Other than that, good work.
First of all, the introduction to this blog post is 10/10. I knew exactly what I was getting into by reading the first paragraph and the information that followed flowed in a logical and well-organized manner. The relevance of the topic is apparent from the jump and the audience is drawn in by the ubiquity immediately. There is no doubt that this is an application that has not only been around for years, but will remain the trusted process for a longtime to come.
The definitions presented throughout the article are clear and the variables are explicitly stated, which I appreciate. I was initially confused by the absolute limits for the amount of ions at the negative electrode being 0% and 100% “both in theory and in reality”. I’m still not sure if I quite understand the limits, but I think explaining why the structure of electrodes degrades would be supplementary (though, not necessary to explain the overall process—which is what this project is about). Are all lithium-ion batteries created equal? Am I damaging my phone’s battery by unnecessarily plugging it in at 80%? Also (this may have been deemed intuitive), stating why lithium ion batteries are chosen over lead acid ones—despite the slight plating issue—may make it more apparent as to why lithium batteries have been deemed the best.
Thank you for unveiling the some of the mystery behind why my phone claims to be dead, but still has enough charge to announce that it needs to be plugged in. The control variable makes sense, because we don’t want to harm the battery by trying to replenish the charge. This article provokes thought and makes me wonder about “low battery mode” on newer phone models and how control can be applied to the battery output as well. The graphics and schematics for the process were very helpful, and though the curves in the graph could have been discussed, they are straightforward and easy to decipher. I’m also curious about chargers that fail, or ones that take super long to charge your phone—not necessary to explain this particular process, but if you were looking to take it a step further, you could!
As you stated this is a technology that has been present for quite some time and manufacturers are always looking to make their product the most competitive. I think another disturbance variable could be the state of the battery itself. For example, if it has undergone many cycles, the 100% is not indicative of the quantity of lithium ions the electrode could originally hold when the phone was fresh out the box. Therefore, fully charged today could me 92% yesterday due to more charging cycles. All in all, this was a great read, accessible to a wide audience, and invoked many questions about an object I am almost literally attached to at the hip.
This idea is not novel; however, it is absolutely useful, such as in climbing, skiing, outdoor adventure and so on, since you cannot always charge your phone when climbing Everest or expedition in skiing to south pole! Furthermore, our phone is omnipotent in daily life; we use it to check our emails, register courses, chat, play games etc. Therefore, limited charge is annoying.
The proposed control scheme makes clear flow and intuitive sense ,The percentage of charge is made by measuring the number of ions available at electrode. Theoretically, the available amount of charge carries can be 100%; however, repeated cycles damage the battery. Therefore, how to charge batteries without damage is so important. Therefore, here, the group can make a feedback loop; when the number of charge carries is saturated, the feeding process will stop automatically. The disturbance variables should be the battery itself, like the variation in number of charge carriers; control variable could be the amount of elections charged by external current or voltage; the manipulate variable could be the intensity of external electrical potential.
The control scheme is viable. In general, the mechanism of charging is by supplying the external current or voltage to separate the charge carriers and built the new internal electrical and chemical potentials. But here, the group an alternative way, they change the constant current flow rate, to protect the battery. However, the most difficult thing is to handle the disturbance viable. The loss of energy is almost in heat, but heat transfer depends on temperature gradient. The gradient is varying all the time, thus the manipulate variable that needs to balance, efficiently, the energy among electrical, chemical, potential and heat is hard to control.
What’s more, the rate of current is hard to measure; I would like to consider that changing rate to energy is easier to measure; alternatively, measuring the amplitude is of current is still easier. The group introduce a new idea, which is to use a feedback loop to compensate the small perturbations in original current.
Finally, I am just wondering, how large the effect and efficiency could be to make the life battery longer by putting it in a good recharging condition. Actually, I would expect to design a new battery with new content of materials, which can change the capacity of battery itself.
I think this post does an excellent job of explaining the basics of a lithium ion battery and how they are implemented for use in a phone. It is also particularly interesting to learn about the absolute min and max charge states should be avoided because they degrade the battery. The introduction does a great job of drawing the reader in, and I think this is in part due to the topic of choice. Cellphones are almost a universal necessity at this point so I think providing the user with practical and important information about how a part of their phone works and how they are designed to ensure safety was a great way to break into the science behind the control mechanism.
The control variable was very clearly defined and explained. I feel much more confident in my understanding of how the lithium battery’s charge state is affected and why it must be controlled. The explanation of the disturbance variables was done well and I had no trouble understanding how fluctuations in current occur due to the plugging and unplugging of other electronics. I do have some questions about the manipulated variable though. I understand that the rate of charge/discharge needs to be controlled and why it needs to be controlled but I feel that the article could do more to explain how exactly it is controlled. Based on the diagram I can discern that the phone sends a signal that alters the charge into the system. What I don’t understand though, is if the phone signals to another part in the phone to slow charge, or if the phone signals the charger itself to slow the charge. Also it is mentioned briefly but how exactly is the rate being manipulated? I understand that resistance is applied to decrease the rate of flow but where is this resistance coming from. I think expanding on these details would help to solidify the article and give a more complete rundown of the control mechanism in place.